GreenBone provides Innovative Solutions for Bone Regeneration

Surgeons

Patients

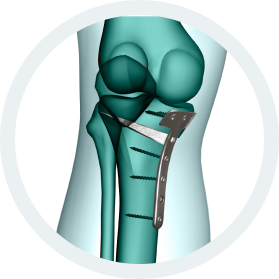
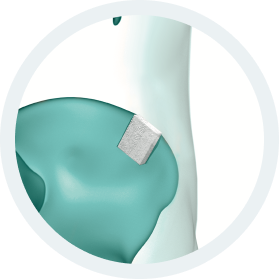



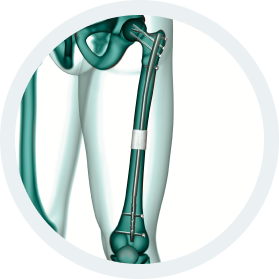

b.Bone Clinical Applications:
- Traumatology
- Bone Augmentation
- Correction of deformity
- Bone Lengthening
- Non union
- Bone loss
- Osteotomy
- Joint revision
b.Bone, several shapes and different sizes
Inspired by Nature
GreenBone Ortho’s mission is to provide innovative, safe and effective solutions for bone regeneration.
GreenBone chose rattan wood because of its intrinsic structure that mimics human bone …

Our technology
The GreenBone Ortho’s technology includes a novel multi-step process able to transform native vegetable structures into inorganic biomaterial.
Our team of scientists and chemists were inspired by Nature to develop an innovative biomimetic and bioactive bone substitute to address unmet clinical needs in bone reconstruction …

Patients
b.Bone acts as a bridge to bone defects, restores bone structures and supports bone healing.
Totally safe and biocompatible vegetal origin, suitable for all patients (1) …
Investors
As main Investors: 3B Future Health Fund, Innogest SGR, CDP Venture Capital, Meta Venture and Italian Angels for Growth (IAG).
















